AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Co2 molecular geometry12/2/2023  Here the valence electrons of Sulfur are used to make Hydrogen stable by donating an electron to both the atoms of Hydrogen. Now place two Hydrogen atoms on both sides of the central atom. Place the Sulphur atom in the middle and arrange its valence electrons around it. But Hydrogen only requires a single electron to become stable as it belongs to Group 1 elements. Sulfur needs eight electrons to fulfill the requirements for Octet Rule. The Lewis structure of H2S is similar to H 2 S. In this compound, both the hydrogen atoms require one electron to make the covalent bond with Sulfur. The Lewis Structure of Hydrogen Sulfide is easy to draw and understand. The Octet Rule of chemistry states that there should be eight electrons in the outer shell of an element for it to be stable. The structure is made based on the Octet Rule. Whereas, the lines represent the bonds in the compound.  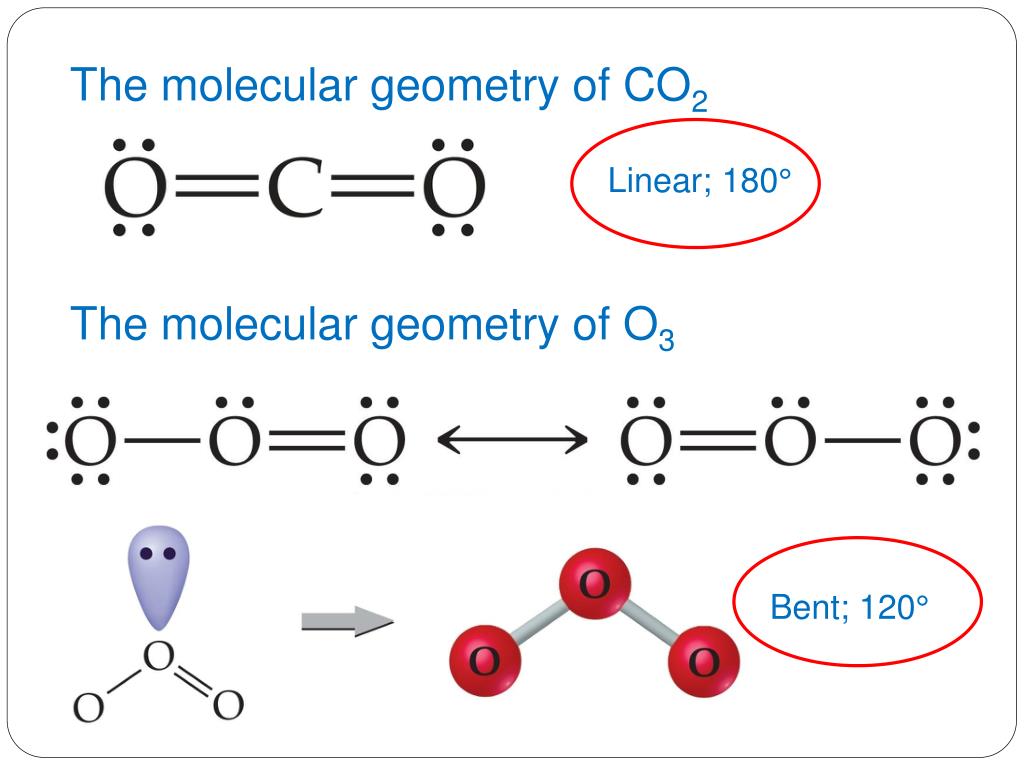
Dots represent the electrons that do not participate in the bond formation. The representation is displayed using dots and lines that represent electrons. Knowing the Lewis structure of a given chemical compound is essential as it provides the necessary information about all other chemical properties of the compound. The Lewis structure of any compound is a structural representation of the valence electrons participating in the formation of bond along with the nonbonding electron pairs. Thus, there are a total of eight valence electrons in H 2 S. Total number of valence electrons in H 2 S Hence there are two valence electrons for Hydrogen atom ( as there are two Hydrogen atoms) There are two atoms of Hydrogen and a single atom of Sulfur in the compound.Įach Hydrogen atom has only one electron which is also its valence electron To know the total number of valence electrons in Hydrogen Sulfide we need to add the valence electrons of both Hydrogen and Sulfur atoms. H2S Polarity Valence electrons of H 2 S.If there are no lone electron pairs on the central atom, the electron pair and molecular geometries are the same.Ĭlick here to see the various molecular geometries.Ĭhoose the correct molecular geometries for the following molecules or ions below. Step 4: The molecular geometry describes the position only of atomic nuclei (not lone electron pairs) of a molecule (or ion). * Lone electron pairs are represented by a line without an atom attached. The table below shows the electron pair geometries for the structures we've been looking at: The regions of electron density will arrange themselves around the central atom so that they are as far apart from each other as possible. 
Use this number to determine the electron pair geometry.Įach bond (whether it be a single, double or triple bond) and each lone electron pair is a region of electron density around the central atom. Step 3: Add these two numbers together to get the regions of electron density around the central atom. Make sure you understand why they are correct. The correct answers have been entered for you. How many lone electron pairs are on the central atom in each of the following Lewis structures? Step 1: Count the number of lone electron pairs on the central atom. Molecular geometry can be predicted using VSEPR by following a series of steps: In other words, the electrons will try to be as far apart as possible while still bonded to the central atom. The theory says that repulsion among the pairs of electrons on a central atom (whether bonding or non-bonding electron pairs) will control the geometry of the molecule. The valence shell electron-pair repulsion theory (abbreviated VSEPR) is commonly used to predict molecular geometry. Lewis structures are very useful in predicting the geometry of a molecule or ion. Many of the physical and chemical properties of a molecule or ion are determined by its three-dimensional shape (or molecular geometry). 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
